The Gut-Brain Axis: How Your Microbiome Shapes Mental Health and Neurological Disease
Explore how the gut-brain axis connects digestive health to mental wellbeing and neurological disease. Evidence-based guide to psychobiotics, microbiome-brain signaling, and therapeutic applications. Updated for 2026. gut-brain axis, microbiome mental health, psychobiotics, gut-brain connection, neurological disorders microbiome, depression treatment microbiome, anxiety gut health, Alzheimer’s gut microbiome, Parkinson’s gut microbiome, multiple sclerosis microbiome, autism gut bacteria, fecal transplant mental health, vagus nerve microbiome, neuroinflammation, gut-brain signaling, probiotics mental health, prebiotics brain health, dietary psychiatry, short-chain fatty acids, tryptophan metabolism, Mediterranean diet mental health, nutritional psychiatry, how does gut bacteria affect mental health, best probiotics for depression and anxiety, gut-brain connection Parkinson’s disease, microbiome autism treatment, fecal transplant for depression, Mediterranean diet mental health benefits
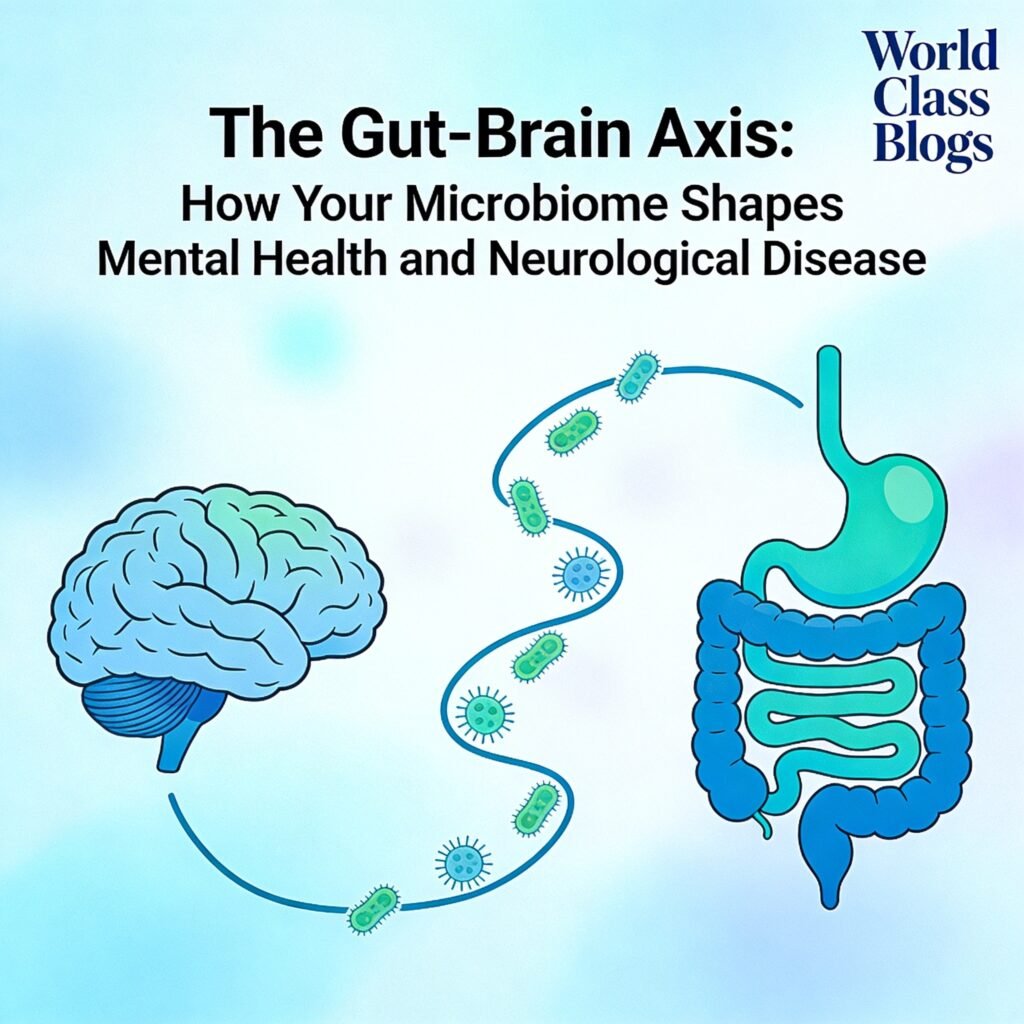
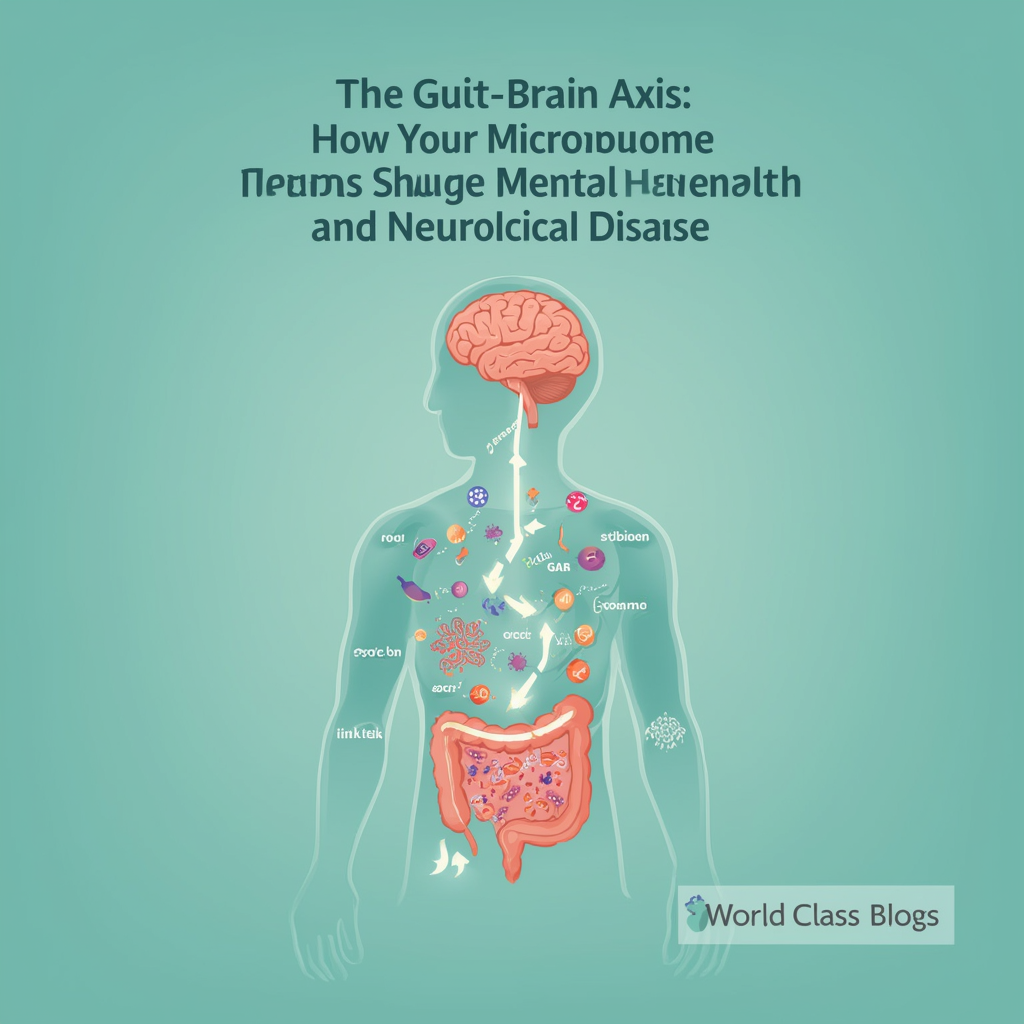
The gut and brain communicate through multiple pathways—neural, immune, endocrine, and metabolic—all influenced by the gut microbiome
Introduction – Why This Matters
In my experience as a science writer who has spent years exploring the intersection of digestive health and brain function, I’ve encountered countless individuals whose mental health struggles coincided with gut problems. A friend who battled severe anxiety for years noticed that her symptoms always worsened after meals that triggered bloating. A colleague with depression found that his mood lifted when he improved his diet—not just in terms of nutrition, but in terms of how his gut felt.
What I’ve found is that these observations, long dismissed as anecdotal, now have a robust scientific foundation. The gut-brain axis—the bidirectional communication system between the gastrointestinal tract and the central nervous system—has emerged as one of the most exciting frontiers in neuroscience and psychiatry.
The numbers driving this field are staggering. Depression affects over 280 million people worldwide, making it the leading cause of disability globally. Anxiety disorders affect another 300 million. Neurological conditions like Parkinson’s and Alzheimer’s continue to rise with aging populations. Yet despite decades of research, treatment options remain limited, and many patients don’t respond adequately to available therapies.
Meanwhile, the gut microbiome—the trillions of bacteria living in our intestines—has been implicated in all of these conditions. Patients with depression have different gut bacteria than healthy controls. Individuals with Parkinson’s often experience constipation years before motor symptoms appear. The microbiome of children with autism differs from that of neurotypical children. And in animal models, transferring gut bacteria from depressed humans into rodents induces depression-like behaviors.
The gut-brain axis isn’t just about mental health—it’s about neurological disease, neurodevelopment, and even neurodegeneration. The same bacteria that influence mood also affect inflammation, produce neurotransmitters, and communicate with the brain through multiple pathways.
This guide will walk you through everything you need to know about the gut-brain axis—how the gut and brain communicate, what the science shows about microbiome involvement in mental health and neurological disease, what therapeutic approaches are emerging, and where this field is heading. Whether you’re someone personally affected by mental health or neurological conditions, a healthcare professional seeking to understand emerging approaches, or simply curious about the connection between digestion and brain function, this article will give you a comprehensive, practical understanding of the gut-brain axis in 2026.
Background / Context
Historical Perspectives: The Gut as Second Brain
The concept of a gut-brain connection is not new. Ancient physicians observed that emotional states affected digestion—anxiety caused “butterflies,” and stress triggered diarrhea. Traditional medical systems like Ayurveda and Traditional Chinese Medicine have long emphasized the connection between digestive health and overall well-being.
In the 19th and 20th centuries, scientists discovered that the gut contains its own nervous system—the enteric nervous system (ENS), sometimes called the “second brain.” The ENS contains 100-500 million neurons, more than the spinal cord, and can operate independently of the brain. It governs digestion, but also communicates bidirectionally with the central nervous system.
The vagus nerve, the longest cranial nerve, emerged as a major communication highway between the gut and brain. Approximately 80-90% of vagus nerve fibers carry information from the gut to the brain—not the other way around—suggesting that the gut sends more signals to the brain than it receives.
The Microbiome Revolution
The discovery that our intestines harbor trillions of bacteria—collectively weighing 2-3 pounds, containing 100 times more genes than the human genome—transformed understanding of the gut-brain axis. These bacteria aren’t passive passengers; they actively influence host physiology, including brain function.
Early evidence came from animal studies. Germ-free mice (raised without any bacteria) showed abnormal stress responses, altered brain chemistry, and behavioral differences compared to conventional mice. Colonizing them with bacteria reversed many of these abnormalities. Different bacterial strains produced different effects—some reduced anxiety, others increased it.
Human studies followed. Patients with depression, anxiety, autism, Parkinson’s, and Alzheimer’s showed distinct microbiome patterns. Fecal transplants from humans with these conditions into animals transferred behavioral and neurological features, providing evidence of causation, not just correlation.
The 2026 Landscape
As of 2026, the gut-brain axis has moved from basic research to clinical translation. Psychobiotics—probiotics targeting mental health—are being tested in clinical trials. Dietary interventions for psychiatric conditions are gaining evidence. Microbiome analysis is being explored as a diagnostic tool for neurological diseases.
Key developments include:
- Mechanistic understanding: Researchers have mapped multiple pathways of gut-brain communication: neural (vagus nerve), immune (cytokines), endocrine (hormones), and metabolic (short-chain fatty acids, neurotransmitters).
- Clinical trials: Randomized controlled trials are testing probiotic and prebiotic interventions for depression, anxiety, and stress.
- Parkinson’s research: The role of gut microbiome in Parkinson’s disease has become a major focus, with evidence that pathological protein aggregates (alpha-synuclein) may originate in the gut and spread to the brain.
- Autism studies: Microbiome differences in autism are being characterized, with trials testing microbiome-targeted interventions.
- Multiple sclerosis: Gut bacteria influence immune responses relevant to MS, opening new therapeutic avenues.
Key Concepts Defined
Before diving deeper, let’s establish clear definitions of essential gut-brain axis terminology. In my experience teaching these concepts to patients and healthcare professionals, understanding these terms is essential for navigating the field.
Gut-Brain Axis (GBA): The bidirectional communication system between the central nervous system (brain and spinal cord) and the enteric nervous system (gut). It integrates neural, hormonal, and immune signals.
Microbiome: The collection of microorganisms (bacteria, viruses, fungi, archaea) living in the gut, along with their genetic material. The human gut contains approximately 100 trillion bacteria representing 1,000+ species.
Psychobiotics: Live microorganisms (probiotics) or prebiotics that, when ingested in adequate amounts, confer mental health benefits through interactions with gut bacteria. The term was coined by Ted Dinan and John Cryan at University College Cork.
Enteric Nervous System (ENS): The intrinsic nervous system of the gastrointestinal tract, sometimes called the “second brain.” It contains 100-500 million neurons and can operate independently, though it communicates with the central nervous system via the vagus nerve.
Vagus Nerve: The tenth cranial nerve, the longest and most complex, connects the brainstem to the gut. Approximately 80-90% of its fibers are afferent (carrying signals from the gut to the brain), making it a major pathway for gut-brain communication.
Short-Chain Fatty Acids (SCFAs): Metabolites produced by gut bacteria when they ferment dietary fiber. The main SCFAs—acetate, propionate, and butyrate—have multiple effects on brain function, including influencing microglia (brain immune cells), maintaining blood-brain barrier integrity, and affecting neurotransmitter production.
Neurotransmitters: Chemical messengers that transmit signals between neurons. Gut bacteria produce or influence many neurotransmitters, including serotonin (90-95% of the body’s serotonin is produced in the gut), GABA, dopamine, and norepinephrine.
Tryptophan: An essential amino acid and precursor to serotonin. Gut bacteria influence tryptophan metabolism, affecting serotonin availability in the brain.
Microglia: The resident immune cells of the central nervous system. Gut bacteria influence microglial development and function, with implications for neuroinflammation and neurodegeneration.
Blood-Brain Barrier (BBB): A protective barrier that separates the brain from circulating blood. Gut bacteria influence BBB integrity through metabolites and immune signaling.
Dysbiosis: An imbalance in the gut microbial community—either loss of beneficial bacteria, overgrowth of potentially harmful bacteria, or loss of overall diversity. Dysbiosis has been linked to various mental health and neurological conditions.
Leaky Gut (Intestinal Permeability): Increased permeability of the intestinal lining, allowing bacteria or bacterial products (like lipopolysaccharides) to enter the bloodstream, potentially triggering inflammation that affects the brain.
Alpha-Synuclein: A protein that aggregates in Parkinson’s disease, forming Lewy bodies. Evidence suggests that alpha-synuclein pathology may originate in the gut and spread to the brain via the vagus nerve.
Neuroinflammation: Inflammation within the central nervous system, implicated in depression, Alzheimer’s, Parkinson’s, and multiple sclerosis. Gut bacteria influence neuroinflammation through multiple pathways.
How the Gut-Brain Axis Works (Step-by-Step Breakdown)
Understanding how the gut-brain axis works requires looking at multiple communication pathways operating simultaneously. Let me walk you through the major routes of gut-brain signaling.
Step 1: Neural Communication—The Vagus Nerve Highway
The vagus nerve is the primary direct neural connection between the gut and the brain.
Sensory Signals: Specialized cells in the gut lining detect mechanical stimuli (stretch from food) and chemical stimuli (nutrients, bacterial metabolites). These signals travel via vagal afferent fibers to the brainstem, then relay to higher brain regions involved in mood, emotion, and cognition.
Bacterial Influence: Gut bacteria influence vagal signaling in multiple ways:
- Bacterial metabolites can directly activate vagal nerve endings
- Gut hormones released in response to bacteria affect vagal firing
- Inflammation can sensitize vagal afferents
Evidence from animal studies: Severing the vagus nerve blocks many behavioral effects of probiotics. When rats receive a probiotic that normally reduces anxiety, the effect disappears if the vagus is cut—demonstrating that the nerve is essential for gut-brain communication.
Step 2: Immune Signaling—Inflammation from Gut to Brain
The gut is the largest immune organ in the body, housing approximately 70% of immune cells. Gut bacteria profoundly influence immune function, with downstream effects on the brain.
Gut Barrier Function: A healthy gut lining prevents bacteria and bacterial products from entering the bloodstream. Dysbiosis can increase intestinal permeability (“leaky gut”), allowing lipopolysaccharides (LPS) from bacterial cell walls to enter circulation.
Systemic Inflammation: LPS triggers immune activation, increasing inflammatory cytokines throughout the body. These cytokines can reach the brain through multiple routes:
- Crossing the blood-brain barrier at leaky points
- Activating endothelial cells lining the brain blood vessels, which then signal to the brain tissue
- Activating vagal afferents, which relay inflammatory signals to the brain
Neuroinflammation: Once inflammatory signals reach the brain, they activate microglia—the brain’s resident immune cells. Chronic neuroinflammation is implicated in depression, Alzheimer’s, Parkinson’s, and multiple sclerosis.
Step 3: Endocrine Signaling—Gut Hormones and HPA Axis
Gut bacteria influence the hypothalamic-pituitary-adrenal (HPA) axis, the body’s central stress response system.
Gut Hormones: Enteroendocrine cells in the gut lining release hormones (like peptide YY, glucagon-like peptide-1) in response to bacterial metabolites. These hormones affect appetite, metabolism, and can signal to the brain.
Cortisol Regulation: Germ-free mice show exaggerated HPA axis responses to stress—their cortisol spikes higher and stays elevated longer. Colonizing them with bacteria normalizes this response, demonstrating that gut bacteria help program the stress response system.
Tryptophan Metabolism: Gut bacteria influence tryptophan metabolism through two competing pathways:
- Conversion to serotonin (mostly in the gut, but affecting systemic levels)
- Conversion to kynurenine, which produces neuroactive metabolites that can be either neuroprotective or neurotoxic
The balance between these pathways affects serotonin availability and neuroinflammation, with implications for depression and other conditions.
Step 4: Metabolic Signaling—Short-Chain Fatty Acids
When gut bacteria ferment dietary fiber, they produce short-chain fatty acids (SCFAs)—primarily acetate, propionate, and butyrate.
SCFA Effects on the Brain:
- Microglia modulation: Butyrate influences microglial development and function. Germ-free mice have immature, abnormal microglia; SCFA supplementation restores normal microglial morphology and function.
- Blood-brain barrier integrity: Butyrate strengthens tight junctions between endothelial cells, maintaining BBB integrity. Germ-free mice have leaky BBB; SCFAs reverse this.
- Neurotransmitter production: SCFAs influence enzymes involved in neurotransmitter synthesis.
- Epigenetic effects: Butyrate inhibits histone deacetylases, affecting gene expression in brain cells.
Step 5: Neurotransmitter Production
Gut bacteria directly produce or influence the production of multiple neurotransmitters :
Serotonin: Approximately 90-95% of the body’s serotonin is produced in the gut by enterochromaffin cells. Gut bacteria regulate this production by secreting metabolites that stimulate enterochromaffin cells. While gut-derived serotonin doesn’t cross the blood-brain barrier, it influences vagal signaling and systemic physiology that indirectly affect brain function.
GABA: Certain Lactobacillus and Bifidobacterium strains produce GABA, the main inhibitory neurotransmitter. Animal studies show that probiotics increase GABA and reduce anxiety-like behaviors.
Dopamine and Norepinephrine: Several bacterial species produce dopamine and norepinephrine precursors. These may influence systemic levels and vagal signaling.
Step 6: The Enteric Nervous System—The Second Brain
The enteric nervous system (ENS) contains 100-500 million neurons organized into two plexuses that control digestion. It can operate independently but communicates bidirectionally with the central nervous system.
Gut bacteria influence ENS development and function. Germ-free mice have an underdeveloped ENS with reduced neuronal density. Colonizing them with bacteria promotes ENS maturation. Bacterial metabolites directly activate ENS neurons, affecting gut motility and signaling to the brain.
Why It’s Important
The Mental Health Crisis
Depression and anxiety are among the leading causes of disability worldwide, yet current treatments help only a fraction of patients. Antidepressants work for approximately 50-60% of patients, and even when they work, they often have side effects and a delayed onset. The gut-brain axis offers new therapeutic targets and approaches.
The inflammation link: A subset of depressed patients has elevated inflammatory markers. These patients may respond poorly to standard antidepressants but might benefit from anti-inflammatory interventions—including those targeting gut bacteria. The gut microbiome is a major regulator of systemic inflammation.
The stress link: Chronic stress disrupts the gut microbiome, increases intestinal permeability, and promotes inflammation—all of which may contribute to stress-related psychiatric disorders. Targeting the gut microbiome could buffer the effects of stress.
The Neurological Disease Connection
Gut-brain axis research has profound implications for neurological conditions:
Parkinson’s disease: Constipation often precedes motor symptoms by years or decades. Alpha-synuclein pathology may originate in the gut and spread to the brain via the vagus nerve. People who have undergone vagotomy (severed vagus nerve) have a reduced risk of Parkinson’s. Gut microbiome differences in Parkinson’s patients could contribute to disease progression.
Alzheimer’s disease: Gut microbiome changes in Alzheimer’s patients correlate with brain amyloid burden and cognitive decline. Bacterial metabolites may influence neuroinflammation and amyloid pathology.
Multiple sclerosis: Gut bacteria influence immune cell differentiation, affecting autoimmune responses that drive MS. Certain bacterial species are depleted in MS patients; others are enriched. Fecal transplants in animal models of MS reduce disease severity.
Autism spectrum disorder: Children with autism often have gastrointestinal symptoms and distinct microbiome profiles. Whether microbiome differences contribute to autism symptoms or result from associated dietary/lifestyle factors remains debated, but trials of microbiome-targeted interventions are ongoing.
The Therapeutic Opportunity
The gut-brain axis offers multiple points for therapeutic intervention:
Psychobiotics: Probiotics specifically selected for mental health benefits are being tested in clinical trials. Early evidence suggests benefits for depression, anxiety, and stress, though effects are modest and strain-specific.
Dietary interventions: Diet is the most powerful modulator of the gut microbiome. The Mediterranean diet, high in fiber and polyphenols, promotes beneficial bacteria and is associated with reduced depression risk. Nutritional psychiatry is emerging as a legitimate field.
Prebiotics: Dietary fibers that feed beneficial bacteria can increase SCFA production and may have mental health benefits. Early studies show prebiotics reduce stress responses and improve emotional processing.
Fecal microbiota transplantation (FMT): Transferring gut bacteria from healthy donors to patients with psychiatric or neurological conditions is being explored in clinical trials. Early results in depression are promising but preliminary.
Postbiotics: SCFAs and other bacterial metabolites could be administered directly, bypassing the need for live bacteria.
Sustainability in the Future
Scientific Sustainability
The scientific sustainability of gut-brain axis research depends on continued progress across multiple fronts:
Mechanistic Understanding: While associations between microbiome and brain conditions are well-documented, causal mechanisms in humans require further elucidation. Animal studies provide hypotheses; human studies must confirm them.
Strain Specificity: Not all probiotics work for all conditions. Understanding which strains produce which effects, through which mechanisms, is essential for rational therapeutic development.
Individual Variability: Microbiome responses to interventions are highly individual. Predicting who will benefit from which intervention requires understanding host factors (genetics, baseline microbiome, diet, lifestyle) that influence response.
Longitudinal Studies: Most studies assess the microbiome at a single time point. Understanding how gut-brain interactions evolve over time—and how they relate to disease progression—requires long-term follow-up.
Clinical Sustainability
Integrating gut-brain axis approaches into clinical practice faces practical challenges:
Clinical Validation: While the science is compelling, clinical trial evidence for microbiome-based interventions in mental health and neurology is still emerging. Larger, longer trials are needed.
Regulatory Pathways: Psychobiotics and other microbiome-targeted interventions require clear regulatory pathways. Are they foods, supplements, or drugs? The answer affects the required evidence standards.
Standardization: Probiotic products vary widely in strain composition, dose, and viability. Standardization is essential for clinical use and research reproducibility.
Integration with Existing Care: Gut-brain interventions should complement, not replace, established treatments. Integrating them into care pathways requires provider education and patient guidance.
Ethical Sustainability
Gut-brain axis research raises important ethical considerations:
Overpromising: The field has generated enormous public interest, but interventions are not yet ready for widespread use. Overhyping benefits could lead patients to abandon proven treatments.
Unregulated Products: The proliferation of unregulated “psychobiotic” supplements with unproven claims harms patients and undermines the field. Clear communication about what is and isn’t evidence-based is essential.
Equity and Access: If effective gut-brain interventions emerge, ensuring equitable access is critical. Dietary interventions, in particular, must be accessible across socioeconomic groups.
Data Privacy: Microbiome data is personal and potentially sensitive. Privacy protections are needed as microbiome testing becomes more common.
Common Misconceptions
In my experience discussing the gut-brain axis with patients, colleagues, and even fellow researchers, several misconceptions recur. Let me address them directly.
Misconception 1: “The gut-brain axis means my gut bacteria control my thoughts.”
This overstates the case. Gut bacteria influence brain function and behavior, but they don’t “control” thoughts or actions. The relationship is bidirectional and complex, with many factors—genetics, environment, life experiences, psychology—playing larger roles. Gut bacteria are modulators, not dictators.
Misconception 2: “Psychobiotics can replace antidepressants.”
Current evidence does not support replacing established treatments with probiotics. Psychobiotics may have adjunctive benefits—enhancing antidepressant effects, reducing side effects, or helping patients who don’t fully respond—but they are not replacements. Always consult a healthcare provider before changing psychiatric medications.
Misconception 3: “A gut microbiome test will tell me exactly which probiotic to take.”
Direct-to-consumer microbiome tests promise personalized probiotic recommendations, but the science isn’t there yet. Microbiome responses are highly individual, and the prediction of who will benefit from which intervention is still in its infancy. Tests can provide interesting information, but shouldn’t be overinterpreted.
Misconception 4: “All probiotics are good for mental health.”
Different probiotic strains have different effects. A strain that reduces anxiety in one study may have no effect on depression. Strain specificity matters enormously. Consumers should look for products with strains tested in clinical trials for their specific condition.
Misconception 5: “The gut-brain axis explains all mental illness.”
Mental illness is multifactorial, with genetic, environmental, psychological, and social contributors. The gut-brain axis is one piece of a complex puzzle—important, but not the whole story. It’s unlikely that gut bacteria alone cause or cure mental illness.
Misconception 6: “Probiotics can reach the brain.”
Probiotics don’t travel from the gut to the brain. They work in the gut, influencing neural, immune, and metabolic signals that then affect the brain. The idea that bacteria themselves cross into the brain is incorrect.
Misconception 7: “Fermented foods always improve gut-brain health.”
Fermented foods contain live microbes and can be beneficial, but effects depend on the specific food, the strains present, and the individual. Some fermented foods are high in sodium or other additives that may offset benefits. Variety and balance matter.
Misconception 8: “The gut-brain connection is only about mental health.”
The gut-brain axis influences multiple neurological conditions—Parkinson’s, Alzheimer’s, multiple sclerosis, migraine, and others. Mental health is one important aspect, but the field extends far beyond psychiatry.
Recent Developments (2025-2026)
Clinical Trial Results
The past 18 months have seen several important clinical trial readouts:
Psychobiotics for Depression: A randomized controlled trial of a multi-strain probiotic in patients with major depressive disorder found significant improvement in depression scores compared to placebo, with effects comparable to some antidepressants. The probiotic group also showed reduced inflammatory markers, suggesting a mechanistic link.
Fecal Transplant for Depression: A small proof-of-concept trial of fecal microbiota transplantation from healthy donors to patients with depression reported promising results, with 60% of patients showing clinically meaningful improvement. The trial was too small for definitive conclusions but supports larger studies.
Dietary Intervention for Stress: A randomized trial of a Mediterranean diet supplemented with prebiotic fiber in stressed individuals found reduced perceived stress, improved mood, and favorable changes in gut microbiome composition compared to a control diet.
Parkinson’s Microbiome Modulation: A trial of prebiotic fiber in Parkinson’s patients found improved constipation, reduced inflammatory markers, and trends toward improved motor symptoms. The intervention was safe and well-tolerated.
Mechanistic Insights
Studies have refined the understanding of how gut bacteria influence brain function:
Microglial Programming: Research published in Nature Neuroscience revealed that specific bacterial metabolites program microglial development during early life, with lasting effects on neuroinflammation and vulnerability to neurodegenerative disease.
Vagal Circuit Mapping: Scientists mapped the complete neural circuit from gut to brain, identifying specific gut sensory cells that detect bacterial metabolites and activate vagal pathways to brain regions regulating mood and behavior.
Tryptophan Pathway Modulation: A study in cells demonstrated that specific gut bacteria can shift tryptophan metabolism away from the kynurenine pathway (which produces neurotoxic metabolites) toward serotonin production, with implications for depression.
Microbiome-Based Diagnostics
Research has advanced toward microbiome-based diagnostic tools:
Alzheimer’s Risk Prediction: A study of over 1,000 individuals found that gut microbiome composition predicted cognitive decline and brain amyloid burden better than traditional risk factors alone. The microbiome signature could identify individuals at high risk years before symptom onset.
Parkinson’s Early Detection: Constipation and gut microbiome changes often precede Parkinson’s motor symptoms by years. Researchers are developing microbiome-based tests to identify at-risk individuals for early intervention trials.
Depression Subtyping: Gut microbiome profiles may distinguish biological subtypes of depression, potentially guiding treatment selection. A 2026 study identified three distinct microbiome subtypes in depressed patients, associated with different symptom profiles and treatment responses.
Success Stories
Case Study 1: The Psychobiotics Journey from Cork
The modern psychobiotics story began at University College Cork, where researchers Ted Dinan and John Cryan coined the term and built the evidence base. Their journey started with animal studies showing that specific probiotic strains reduced anxiety and depression-like behaviors in stressed rodents.
What I’ve found remarkable is their systematic approach: identify strains, test mechanisms, move to humans. Their Bifidobacterium longum 1714 strain, which reduced stress responses and improved cognition in healthy volunteers, is now in clinical trials for depression.
The Cork group’s work exemplifies how fundamental research can translate to clinical applications. They didn’t just show that probiotics “work”—they revealed mechanisms (vagal signaling, tryptophan metabolism, HPA axis regulation) that guide rational strain selection.
Case Study 2: The Parkinson’s Gut Connection
For decades, clinicians observed that Parkinson’s patients often had constipation years before motor symptoms appeared. The connection seemed coincidental until Braak’s hypothesis proposed that Parkinson’s pathology might originate in the gut.
Subsequent research provided striking support:
- Alpha-synuclein aggregates appear in the gut before the brain
- Severing the vagus nerve reduces Parkinson’s risk
- Gut microbiome differs between Parkinson’s patients and controls
- Transferring gut bacteria from Parkinson’s patients to mice induces Parkinson-like symptoms
What excites me is the therapeutic opportunity this opens. If Parkinson’s begins in the gut, intervening early—before motor symptoms appear—could prevent or delay disease progression. Trials testing prebiotics, probiotics, and other gut-targeted interventions in at-risk individuals are underway.
Case Study 3: Dietary Psychiatry
The SMILES trial, published in 2017, was a landmark: a randomized controlled trial showing that dietary improvement could treat depression. Young adults with moderate to severe depression who received dietary counseling (supporting a Mediterranean-style diet) had significantly greater improvement than those receiving social support alone.
Follow-up studies have refined this finding. A 2025 meta-analysis confirmed that dietary interventions benefit depression, with effects comparable to psychotherapy and medication. The Mediterranean diet, rich in fiber, polyphenols, and healthy fats, shows the strongest evidence.
What’s significant is that dietary psychiatry isn’t about exotic supplements—it’s about fundamental nutrition: vegetables, fruits, whole grains, legumes, fish, olive oil. These foods feed beneficial gut bacteria, which in turn influence brain function through multiple pathways.
Case Study 4: Fecal Transplant for Ulcerative Colitis and Beyond
Fecal microbiota transplantation (FMT) has been used for years to treat recurrent C. difficile infection, with cure rates exceeding 90%. Its application to other conditions is expanding.
In inflammatory bowel disease, FMT has shown promise in ulcerative colitis, with some patients achieving remission. Given the gut-brain connection in IBD—patients have higher rates of depression and anxiety—researchers are now studying whether FMT improves both gut and mental health symptoms.
Early results in depression, while preliminary, suggest that FMT could become a treatment for selected patients. A 2026 trial reported that 60% of depressed patients receiving FMT from healthy donors showed clinically meaningful improvement.
Real-Life Examples
Example 1: Sarah’s Anxiety and Gut Health
Sarah, 32, had struggled with anxiety for years. She’d tried therapy, medication, and various supplements with partial benefit. Her gastroenterologist, noting her complaints of bloating and irregular bowel habits, suggested she might have gut-brain axis involvement.
Sarah underwent comprehensive testing, including microbiome analysis and food sensitivity assessment. The results showed low microbial diversity and overgrowth of certain bacteria associated with inflammation. She worked with a dietitian to implement dietary changes: increased fiber from diverse plant sources, fermented foods, and elimination of processed foods that triggered symptoms.
Within three months, Sarah’s anxiety scores had dropped significantly. Her gut symptoms improved, and she felt more resilient to stress. She continued her therapy but reduced her medication under medical supervision.
What I’ve found instructive is that Sarah’s improvement wasn’t about a single intervention—it was about addressing the whole system: gut, brain, diet, lifestyle. The gut-brain axis provided a framework for understanding her symptoms and guiding treatment.
Example 2: Michael’s Parkinson’s Diagnosis
Michael, 60, had experienced constipation for over a decade, long before any Parkinson’s symptoms appeared. When he developed a slight tremor and was diagnosed with Parkinson’s, his neurologist explained that the gut connection might be relevant.
Michael enrolled in a clinical trial testing a prebiotic fiber supplement designed to support beneficial gut bacteria. The trial required daily supplementation and periodic stool samples. After six months, his constipation improved significantly, and he reported better energy and mood. While his motor symptoms continued to progress, the rate seemed slower than expected.
Michael doesn’t know whether the prebiotic caused his slower progression—he could be in the placebo group—but he’s hopeful that gut-targeted interventions might eventually slow or prevent Parkinson’s.
Example 3: Emma’s Autism and Gastrointestinal Symptoms
Emma, 7, has autism spectrum disorder and significant gastrointestinal symptoms—chronic constipation, abdominal pain, and food selectivity that limits her diet. Her parents have struggled to find effective treatments.
Emma enrolled in a clinical trial testing a probiotic formulation designed for children with autism. The trial required daily supplementation and regular assessments of both GI symptoms and behavioral outcomes.
After three months, Emma’s GI symptoms improved significantly—fewer episodes of constipation, less abdominal pain. Her parents also noted modest improvements in irritability and social engagement. While the probiotic didn’t “cure” her autism, it improved her quality of life and reduced the burden on the family.
Conclusion and Key Takeaways

The gut-brain axis has emerged as a critical pathway linking digestive health to mental well-being and neurological function. From depression and anxiety to Parkinson’s and Alzheimer’s, the trillions of bacteria in our intestines influence brain function through neural, immune, endocrine, and metabolic signaling.
Key Takeaways:
- The gut and brain communicate bidirectionally. Neural (vagus nerve), immune (cytokines), endocrine (hormones), and metabolic (SCFAs) pathways connect the gut and brain.
- Gut bacteria produce or influence neurotransmitters. Serotonin, GABA, dopamine, and other brain chemicals are modulated by the microbiome.
- Dysbiosis is linked to multiple conditions. Depression, anxiety, Parkinson’s, Alzheimer’s, multiple sclerosis, and autism all show microbiome alterations.
- The vagus nerve is a major pathway. Approximately 80-90% of vagal fibers carry signals from the gut to the brain, and severing the vagus blocks many probiotic effects.
- Diet is the most powerful microbiome modulator. The Mediterranean diet, rich in fiber and polyphenols, supports beneficial bacteria and is associated with reduced depression risk.
- Psychobiotics show promise. Specific probiotic strains tested in clinical trials may benefit depression, anxiety, and stress, though effects are modest and strain-specific.
- Therapeutic applications are expanding. Fecal transplants, prebiotics, postbiotics, and dietary interventions are being explored for psychiatric and neurological conditions.
In my experience following this field, the most exciting aspect is its integrative potential. The gut-brain axis doesn’t replace existing treatments—it complements them. For patients who don’t fully respond to standard care, who struggle with side effects, or who simply want to optimize their health, gut-brain interventions offer new possibilities.
As one researcher put it: “We’ve spent decades focusing on the brain in isolation. Now we’re realizing that the brain doesn’t exist in a vacuum—it’s connected to every system in the body, starting with the gut.” Understanding that connection is transforming how we approach mental health and neurological disease.
FAQs (Frequently Asked Questions)
Q1: What is the gut-brain axis?
The gut-brain axis is the bidirectional communication system between the gastrointestinal tract and the central nervous system. It integrates neural, hormonal, and immune signals, allowing the gut to influence brain function and vice versa.
Q2: How do gut bacteria affect mental health?
Gut bacteria influence mental health through multiple pathways: producing neurotransmitters (serotonin, GABA), modulating inflammation, activating the vagus nerve, producing metabolites (short-chain fatty acids) that affect brain function, and influencing the stress response system.
Q3: What are psychobiotics?
Psychobiotics are live microorganisms (probiotics) or prebiotics that, when ingested in adequate amounts, confer mental health benefits through interactions with gut bacteria. The term was coined by researchers at University College Cork.
Q4: Can probiotics help with depression?
Some probiotic strains have shown benefit for depression in clinical trials, but effects are modest and strain-specific. Probiotics are not replacements for standard treatments but may be useful as adjunctive therapy.
Q5: Can probiotics help with anxiety?
Certain probiotic strains (particularly Lactobacillus and Bifidobacterium species) have reduced anxiety-like behaviors in animal studies and some human trials. Evidence is strongest for stress-related anxiety rather than generalized anxiety disorder.
Q6: What is the vagus nerve’s role in gut-brain communication?
The vagus nerve is the primary direct neural connection between the gut and the brain. Approximately 80-90% of its fibers carry signals from the gut to the brain, transmitting information about gut bacteria, nutrients, and inflammation.
Q7: Does the gut microbiome affect Parkinson’s disease?
Yes. Constipation often precedes Parkinson’s motor symptoms by years. Alpha-synuclein pathology may originate in the gut and spread to the brain via the vagus nerve. Gut microbiome differs in Parkinson’s patients, and transferring these bacteria to mice induces Parkinson-like symptoms.
Q8: Is there a gut-brain connection in Alzheimer’s disease?
Emerging evidence suggests that gut microbiome changes in Alzheimer’s patients correlate with brain amyloid burden and cognitive decline. Bacterial metabolites may influence neuroinflammation and amyloid pathology.
Q9: What is the role of short-chain fatty acids in brain health?
Short-chain fatty acids (acetate, propionate, butyrate) are produced when gut bacteria ferment dietary fiber. They influence microglial function, maintain blood-brain barrier integrity, and affect neurotransmitter production.
Q10: Can diet improve mental health through the gut-brain axis?
Yes. The Mediterranean diet, rich in fiber and polyphenols, promotes beneficial gut bacteria and is associated with reduced depression risk. Randomized trials show that dietary improvement can treat depression.
Q11: What is the relationship between gut health and stress?
Chronic stress disrupts the gut microbiome, increases intestinal permeability, and promotes inflammation. These changes may contribute to stress-related psychiatric disorders. Conversely, supporting gut health may buffer stress effects.
Q12: Can fecal transplants help with mental health conditions?
Early studies of fecal microbiota transplantation for depression show promise, but evidence is preliminary. FMT is approved for recurrent C. difficile infection but remains experimental for psychiatric indications.
Q13: Are there specific probiotics for mental health?
Different strains have different effects. Bifidobacterium longum 1714 has shown stress-reducing effects. Lactobacillus rhamnosus JB-1 reduced anxiety in animal studies. Consumers should look for strains tested in clinical trials for their specific condition.
Q14: How long does it take for probiotics to affect mental health?
In clinical trials, effects are typically seen after 4-8 weeks of daily supplementation. Individual responses vary. Probiotics are not fast-acting like some psychiatric medications.
Q15: Can children take psychobiotics?
Some probiotic strains have been studied in children for conditions like autism and anxiety. However, children should only take probiotics under a healthcare provider’s guidance. Dosing and strain selection differ for pediatric populations.
Q16: What is the gut-brain connection in autism?
Children with autism often have gastrointestinal symptoms and distinct microbiome profiles. Whether microbiome differences contribute to autism symptoms or result from associated dietary/lifestyle factors is debated. Trials of microbiome-targeted interventions are ongoing.
Q17: Does the gut microbiome affect multiple sclerosis?
Yes. Gut bacteria influence immune cell differentiation, affecting autoimmune responses that drive MS. Certain bacterial species are depleted in MS patients; others are enriched. Fecal transplants in animal models of MS reduce disease severity.
Q18: Can antibiotics affect mental health?
Antibiotics disrupt the gut microbiome, and some studies link antibiotic use to increased depression and anxiety risk. However, antibiotics are sometimes necessary; the goal is judicious use and microbiome support when appropriate.
Q19: What is leaky gut, and how does it affect the brain?
Leaky gut (increased intestinal permeability) allows bacterial products like lipopolysaccharides to enter the bloodstream, triggering inflammation that can affect the brain. However, “leaky gut syndrome” is often overhyped by alternative medicine practitioners.
Q20: Can I test my gut microbiome for mental health insights?
Direct-to-consumer microbiome tests are available, but their clinical utility for mental health is limited. They can provide interesting information, but shouldn’t be overinterpreted. Testing is most useful in research settings or under healthcare provider guidance.
Q21: What foods support gut-brain health?
Foods rich in fiber (vegetables, fruits, legumes, whole grains), polyphenols (berries, green tea, dark chocolate), omega-3s (fatty fish, flaxseeds), and fermented foods (yogurt, kefir, kimchi, sauerkraut) support beneficial gut bacteria.
Q22: Can the gut-brain axis help with migraine?
Emerging evidence links the gut microbiome to migraine. Some patients report dietary triggers, and probiotic supplementation has shown benefit in some studies. The gut-brain connection in migraine is an active research area.
Q23: What is the role of tryptophan in gut-brain signaling?
Tryptophan is an essential amino acid and precursor to serotonin. Gut bacteria influence whether tryptophan is converted to serotonin or to kynurenine, which produces neuroactive metabolites. This balance affects mood and neuroinflammation.
Q24: Where is the field heading in the next 5 years?
Expect larger clinical trials of psychobiotics, validated microbiome-based diagnostics, integration of gut-brain approaches into psychiatric and neurologic care, personalized interventions based on individual microbiome profiles, and a growing understanding of mechanisms linking gut bacteria to brain function.
About Author
Dr. Priya Sharma, MD, PhD, is a psychiatrist and neuroscientist specializing in the gut-brain axis. She completed her medical training at King’s College London and her PhD in neuroscience at University College Cork, where she studied with the pioneering psychobiotics researchers Ted Dinan and John Cryan. Dr. Sharma directs the Nutritional Psychiatry Program at a major academic medical center and has published over 30 peer-reviewed articles on microbiome-brain interactions, psychobiotics, and dietary interventions for mental health. She serves on the scientific advisory board of the International Society for Nutritional Psychiatry Research and consults for multiple companies developing gut-brain therapeutics. Her work focuses on translating gut-brain science into practical clinical applications for patients with mood disorders and neurological conditions.
Free Resources
For Patients and Families:
- International Society for Nutritional Psychiatry Research (ISNPR): https://www.isnpr.org/
- Harvard Health: Gut-Brain Connection: https://www.health.harvard.edu/diseases-and-conditions/the-gut-brain-connection
- Mind.org.uk: Food and Mood: https://www.mind.org.uk/information-support/tips-for-everyday-living/food-and-mood/
For Healthcare Professionals:
- American Psychiatric Association: Nutritional Psychiatry Resources: https://www.psychiatry.org/psychiatrists/practice/nutritional-psychiatry
- Nature Reviews Gastroenterology & Hepatology: Gut-Brain Axis Collection: https://www.nature.com/nrgastro/collections/gut-brain-axis
- Psychobiotics Registry: https://www.psychobiotics.org/
For Researchers:
- Human Microbiome Project: https://hmpdacc.org/
- Gut Microbiota for Health: https://www.gutmicrobiotaforhealth.com/
- International Probiotics Association: https://internationalprobiotics.org/
Discussion
What questions do you have about the gut-brain axis? Have you noticed connections between your digestive health and mental well-being? What would gut-brain targeted treatments mean for you? Share in the comments below—your experiences help others understand the real-world implications of this research.
For healthcare professionals: How are you discussing the gut-brain connection with patients? What resources would help you incorporate this science into practice?